EyeBOX / TBI Testing







How EyeBOX Works
EyeBOX captures 100,000 data points based on subtle eye movements and cranial nerve function, generating the BOX score and report indicating the likelihood of concussion.

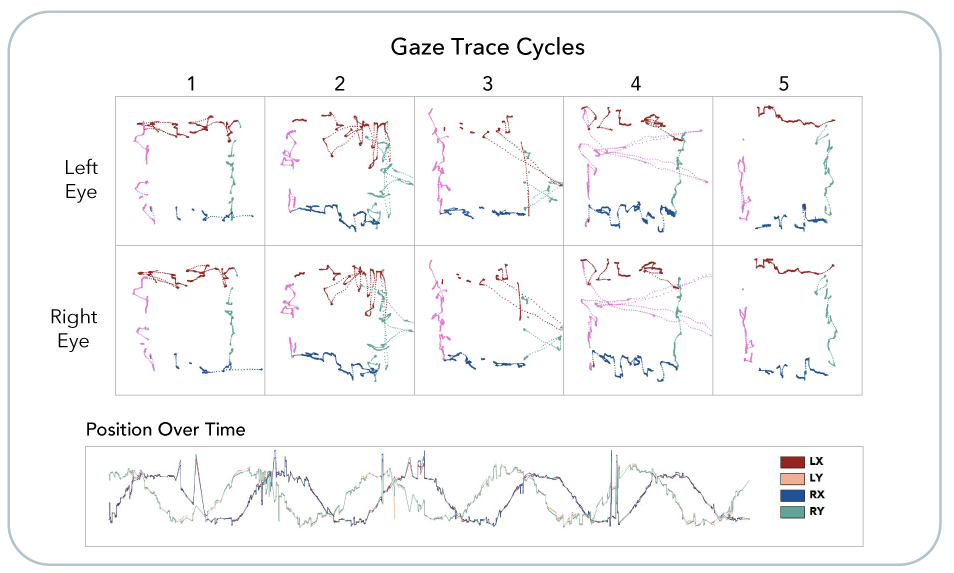
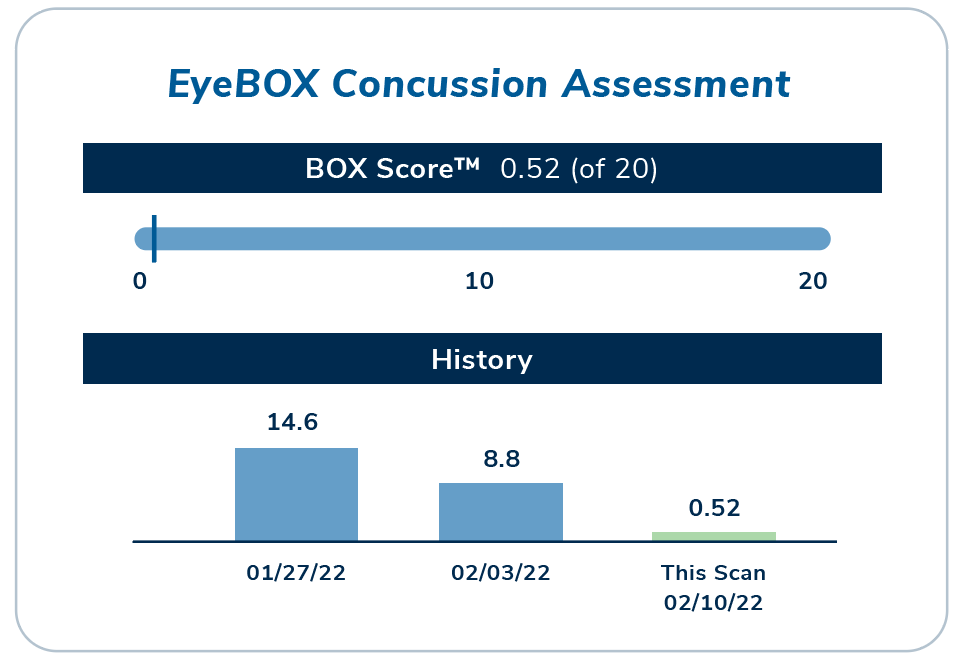







EyeBOX captures 100,000 data points based on subtle eye movements and cranial nerve function, generating the BOX score and report indicating the likelihood of concussion.



